Key secondary endpoint: overall survival
At a median follow-up of 39 months, KISQALI demonstrated a significant OS benefit in the ITT population. mOS was NR in the KISQALI +
fulvestrant arm vs 40.0 months in the placebo + fulvestrant arm (HR=0.72; 95% CI: 0.57–0.92; p=0.00455).2
In an exploratory analysis, at a median follow-up of 71 months, 1L KISQALI + fulvestrant reported a 15.8-month increase in mOS compared
with placebo + fulvestrant (67.6 months vs 51.8 months, respectively; HR=0.67; 95% CI: 0.50–0.90).1
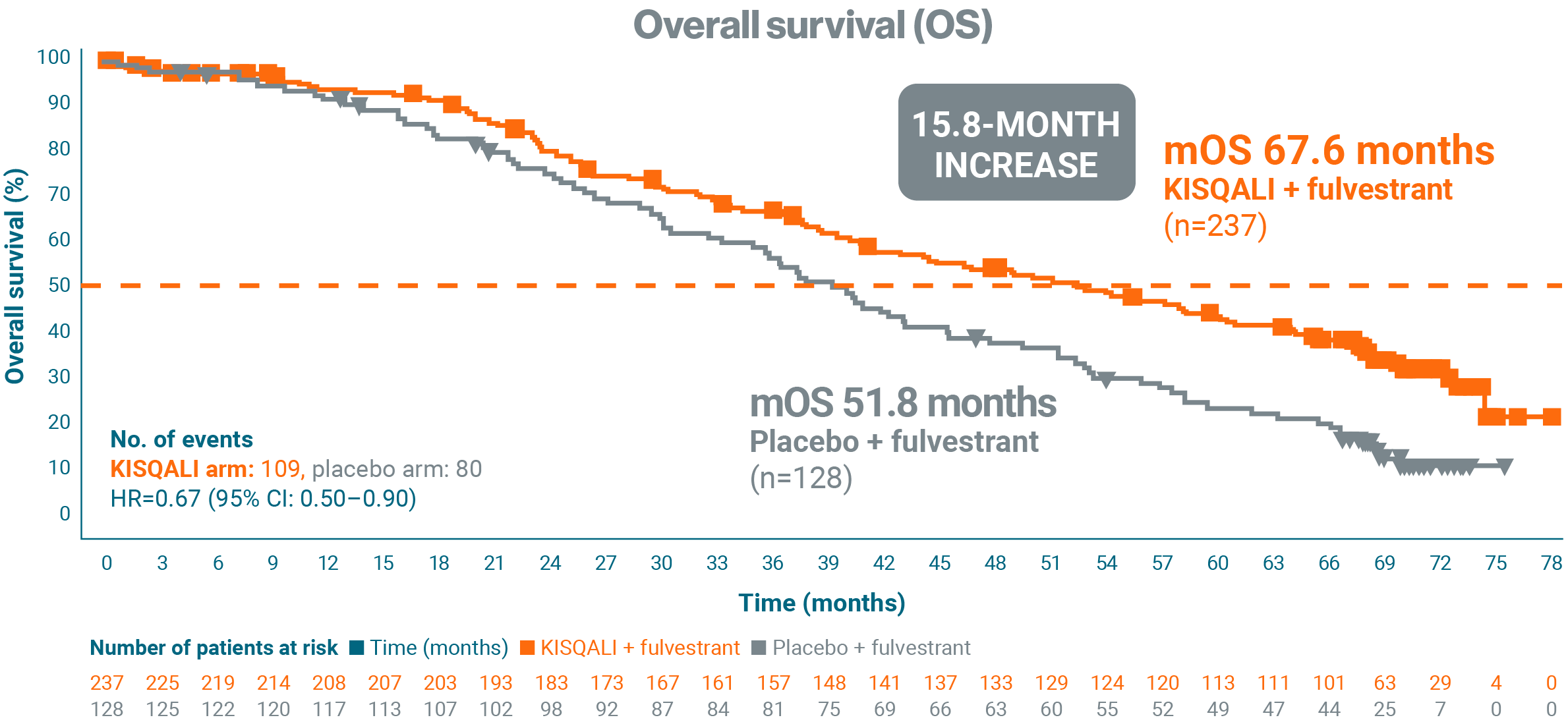
This analysis was not prespecified and
results are observational in nature;
as such, there was no prespecified
statistical procedure controlling
for type 1 error. Results should be
interpreted with caution due to the
limitations of exploratory analyses.1
Adapted from Neven P, et al. 2023.1
In an exploratory analysis, 1L KISQALI + fulvestrant was observed to demonstrate significant OS vs fulvestrant + placebo in postmenopausal patients1
1L, first-line; 2L, second-line; CI, confidence interval; HR, hazard ratio; ITT, intention-to-treat; mOS, median overall survival; NR, not reached.
References
1. Neven P, et al. Breast Cancer Res 2023;25–103.
2. Slamon DJ, et al. N Engl J Med 2020;382(6):514–524.
Sub-analysis of overall survival by line of treatment1
In an exploratory analysis, 2L KISQALI + fulvestrant was observed to have a longer mOS in postmenopausal patients* vs placebo + fulvestrant.1
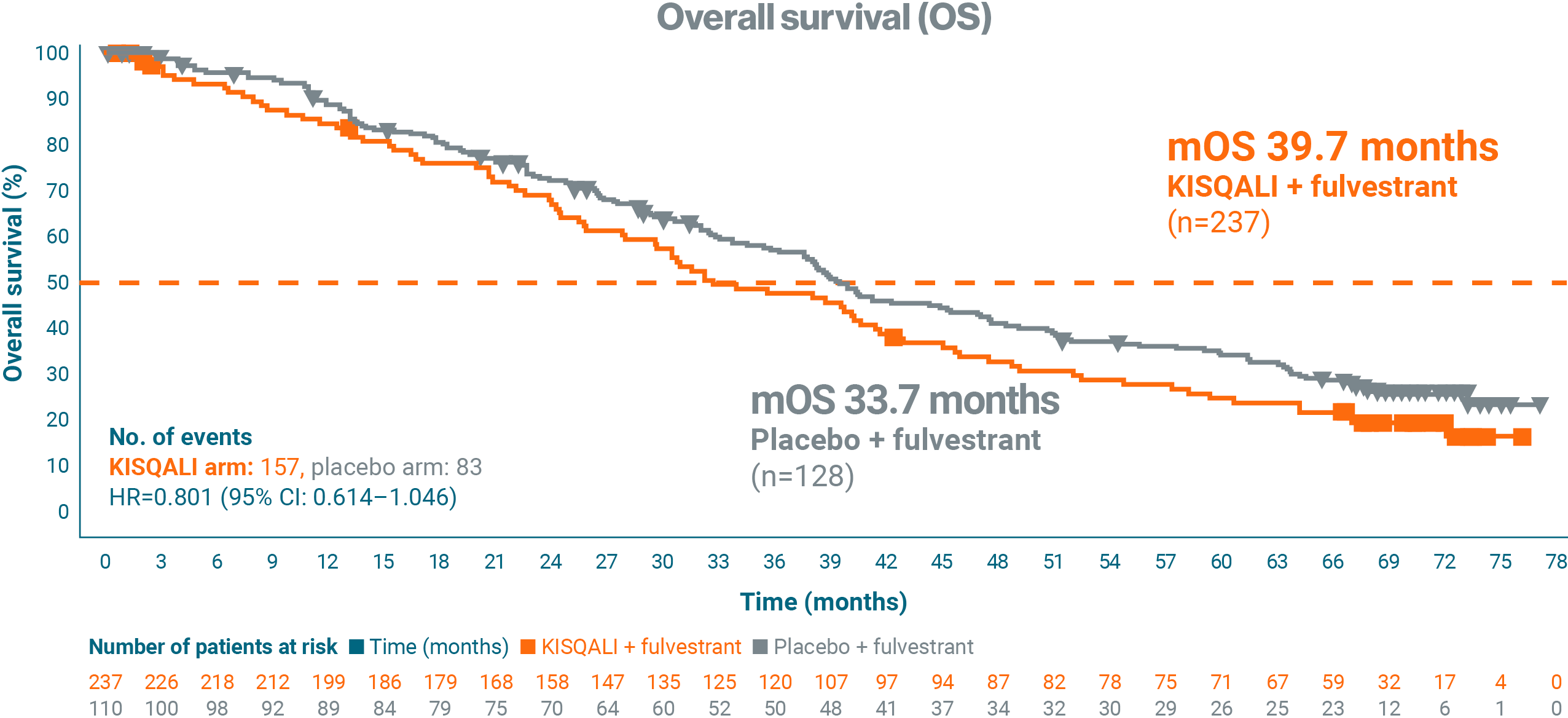
Adapted from Neven P, et al. 2023.1
This was an exploratory OS analysis with an extended median follow-up of 71 months.1
*Early relapse or one prior ET for aBC.1
2L, second line; aBC, advanced breast cancer; CI, confidence interval; ET, endocrine therapy; HR, hazard ratio; mOS, median overall survival.
Reference
1. Neven P, et al. Breast Cancer Res 2023;25:103.
MONALEESA-3