Primary endpoint: progression-free survival
In the primary analysis ITT population, at a median follow-up of 20.4 months, an mPFS of 20.5 months was
achieved in
the KISQALI + fulvestrant arm vs 12.8 months in the placebo + fulvestrant arm; HR=0.59;
95% CI: 0.48–0.73; p<0.001.1
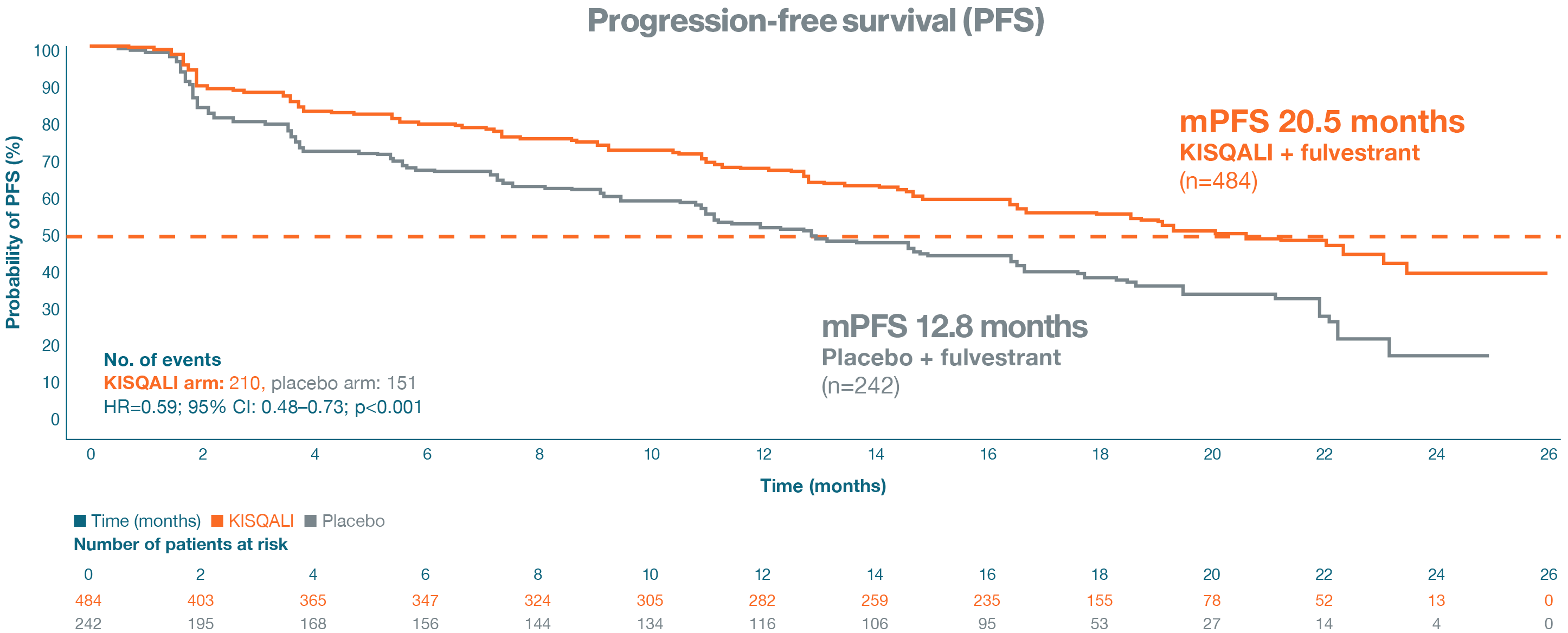
Adapted from Slamon DJ, et al. 2018.1
CI, confidence interval; HR, hazard ratio; ITT, intention-to-treat; mPFS, median progression-free survival.
Reference
1. Slamon DJ, et al. J Clin Oncol 2018;36(24):2465–2472.