Primary endpoint: progression-free survival
KISQALI + AI provides a statistically significant mPFS benefit of 9.3 months vs placebo + AI. In the
primary analysis
ITT population, at a median follow-up of 26.4 months, KISQALI + AI had an mPFS
of 25.3 months vs 16.0 months
in the placebo + AI arm; HR=0.57; 95% CI: 0.46–0.70;
p=9.63x10-8.1
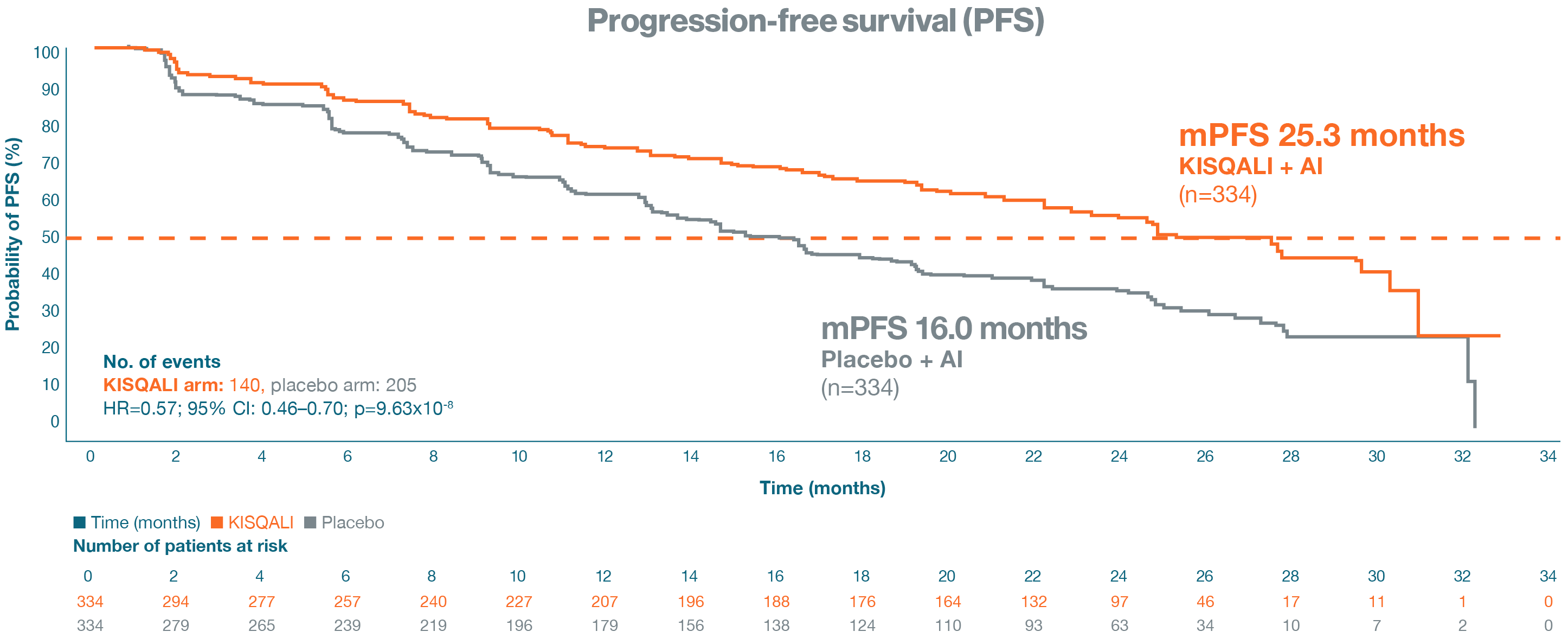
Adapted from Hortobagyi GN, et al. 2018.1
AI, aromatase inhibitor; CI, confidence interval; HR, hazard ratio; ITT, intention-to-treat; mPFS, median progression-free survival.
Reference
1. Hortobagyi GN, et al. Ann Oncol 2018;29:1541–1547.