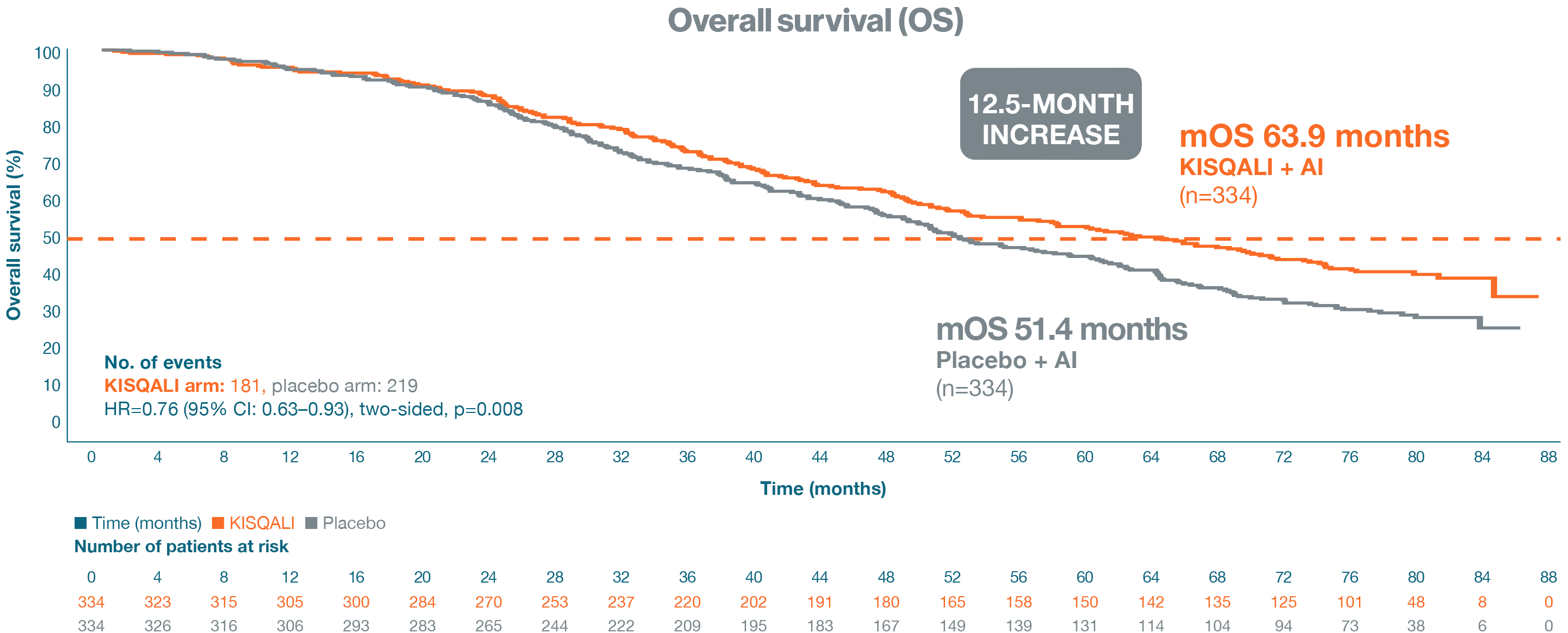
Key secondary endpoint: overall survival
At median 80-month follow-up, KISQALI + AI achieved mOS of 63.9 months vs 51.4 months with placebo + AI (HR=0.76; 95% CI: 0.63–0.93; p=0.008).1
Adapted from Hortobagyi GN, et al. 2022.1
1L KISQALI + AI increased mOS by >1 year (12.5 months) vs placebo + AI in postmenopausal patients1
1L, first line; AI, aromatase inhibitor; CI, confidence interval; HR, hazard ratio; mOS, median overall survival.
Reference
1. Hortobagyi GN, et al. N Engl J Med 2022;386(10):942–950.