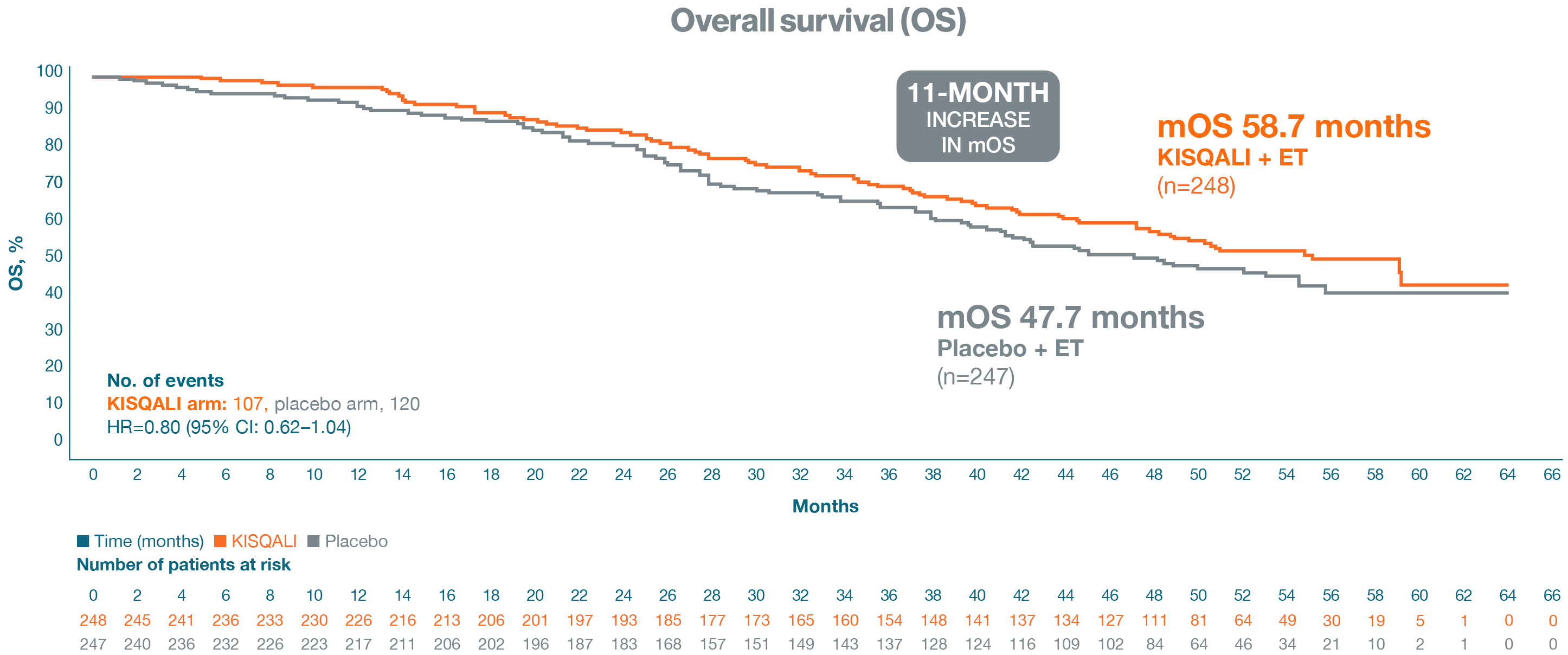
Secondary endpoint: Overall survival
This was an exploratory OS analysis for the NSAI subgroup with an extended median follow-up of 54 months
Adapted from Lu Y-S, et al. 2022.1
In an exploratory analysis, 1L KISQALI + ET was observed to increase mOS by 11 months vs placebo + ET in pre/perimenopausal patients1
ET refers to LHRH plus either NSAI or tamoxifen.
KISQALI is not recommended to be used in combination with tamoxifen.2
1L, first-line; CI, confidence interval; ET, endocrine therapy; HR, hazard ratio; LHRH, luteinising hormone-releasing hormone; mOS, median overall survival; NSAI, non-steroidal aromatase inhibitor; OS, overall survival.
References
1. Lu Y-S, et al. Clin Cancer Res 2022;28(5):851–859.
2. KISQALI® (ribociclib) Summary of Product Characteristics.