Primary endpoint: Progression-free survival
At a median follow-up of 19.2 months, mPFS was 23.8 months (95% CI: 19.2–not reached) in the KISQALI + ET group vs 13.0 months (95% CI: 11.0–16.4) in the placebo + ET group (HR=0.55; 95% CI: 0.44–0.69; p<0.0001).1
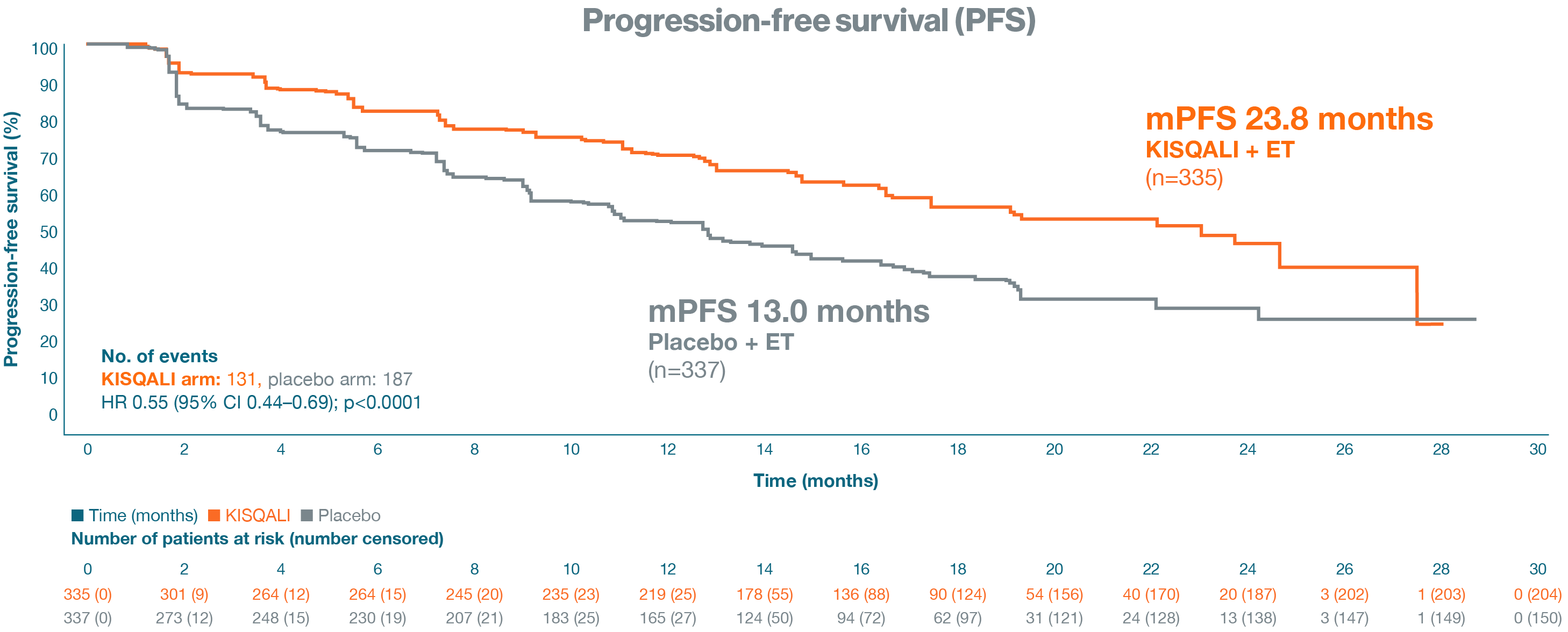
Adapted from Tripathy D, et al. 20181
ET refers to LHRH plus either NSAI or tamoxifen.
KISQALI is not recommended to be used in combination with tamoxifen.2
CI, confidence interval; ET, endocrine therapy; HR, hazard ratio; LHRH, luteinising hormone-releasing hormone; mPFS, median progression-free survival; mPFS2, progression-free survival following receipt of subsequent therapy; NSAI, non‑steroidal aromatase inhibitor.
References
1. Tripathy D, et al. Lancet Oncol 2018;19(7):904–915.
2. KISQALI® (ribociclib) Summary of Product Characteristics.